
In accordance with idarucizumab’s approved indication, enrolled participants were prescribed dabigatran, then presented with uncontrolled or life-threatening bleeding (group A, n = 301) or the need for urgent surgery or procedure (group B, n = 202). 
The full cohort analysis of this study was published in August 2017. 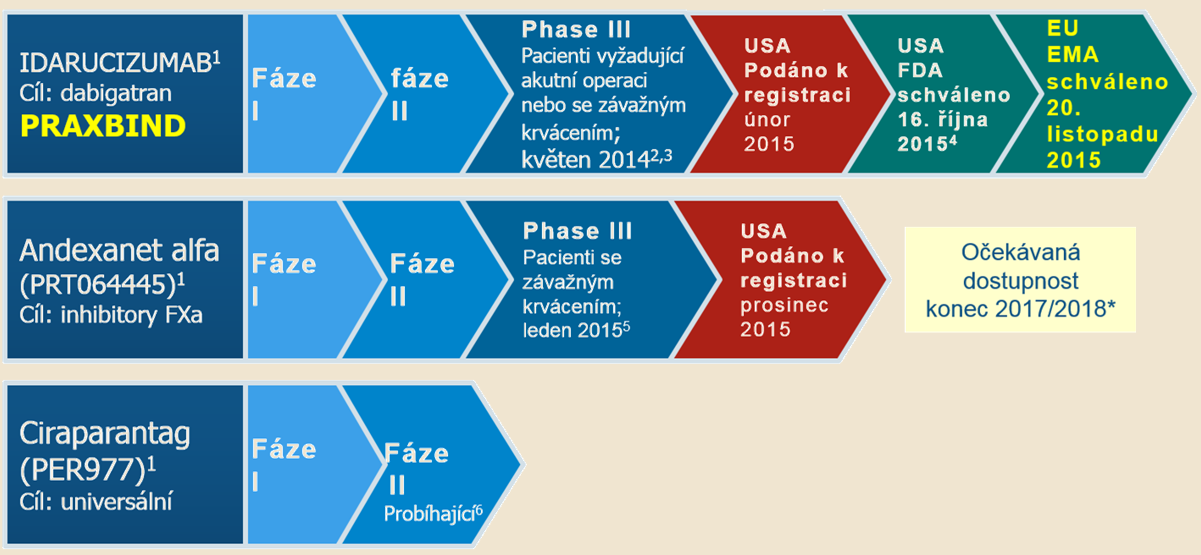
This medicine has been shown to rapidly and completely reverse the anticoagulant effects of dabigatran in the open-label, phase III RE‑VERSal Effects of idarucizumab on Active Dabigatran (RE‑VERSE AD) study. Clinical data to support the use of idarucizumab are still limited.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
